Lightning Produces Molecules that Clean Greenhouse Gases from the Atmosphere
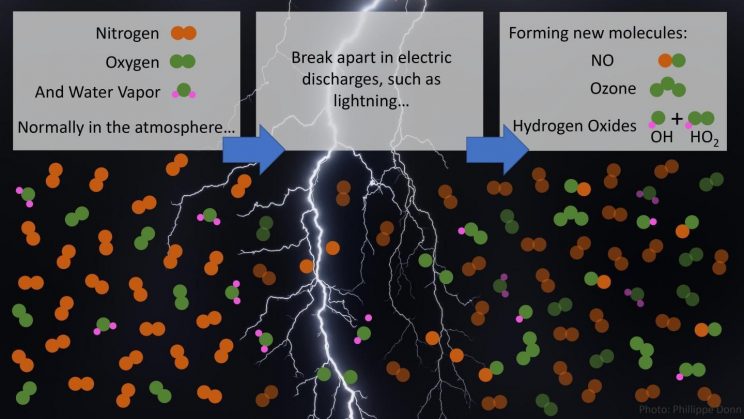
Nitrogen, oxygen and water vapor molecules are broken apart by lightning and associated weaker electrical discharges, generating the reactive gases NO, O3, HO2 and the atmosphere’s cleanser, OH. Credit: Jena Jenkins, Penn State
Lightning produces molecules that can “clean” the atmosphere, according to research published this month in Science (Brune et al., 2021) and authored by a number of NOAA research scientists. The research demonstrates not only that lightning produces hydroxyl (OH) and hydroperoxyl (HO2), but also produces amounts much greater than expected due to weaker, subvisible electrical discharges. Researchers knew that lightning creates ions and new molecules including OH and HO2, but the amounts observed in this experiment were up to 1000 times what researchers had previously observed in the troposphere.
Lightning, and even smaller electric discharges in the atmosphere, creates ozone (O3) and two oxidizing chemicals: hydroxyl (OH) and hydroperoxyl (HO2). The OH and HO2 molecules dominate atmospheric oxidation chemistry that removes the majority of trace gases including some greenhouse gases (GHG), particularly methane, which mitigates the GHG contribution to climate change.
This research in Science came from airborne measurements made during the Deep Convective Clouds and Chemistry (DC3) field campaign in 2012. A range of instrumentation collected over 100 atmospheric constituents both airborne and on the ground-based measurements by using a NASA DC-8 aircraft, a Gulfstream-V from NCAR, satellites, ground-based radar and lightning antenna stations.



